Updated September 2024
Viral infections are the leading cause of gastroenteritis globally and may also cause enterically transmitted hepatitis and illness after migrating from the human intestine to other organs. Various viruses have been implicated in foodborne illness, with three viruses: norovirus (NoV), hepatitis A virus (HAV), and hepatitis E virus (HEV) causing the most significant burden of foodborne illness and outbreaks. Rotavirus, sapovirus, enterovirus, astrovirus and enteric adenovirus have also been associated with foodborne transmission but are currently not considered to be as significant.
Annually, NoV causes an estimated 125 million cases of foodborne illness with 35,000 deaths recorded globally. HAV is estimated to cause 14 million cases of foodborne illness with 28,000 deaths recorded globally. There are currently no global estimates for HEV available1. NoV and HAV primarily originate from the human intestine. They are excreted in high numbers through faeces or vomit. Symptoms are typically nausea, vomiting, diarrhoea and abdominal pain. In the case where a person has a hepatitis A or E infection, jaundice is also a common symptom. HEV is a zoonotic pathogen primarily associated with contaminated pork products. These viruses' low infectious dose and robust survival in food and various environments facilitate the spread of viral infections. Fresh produce may be extrinsically contaminated during primary production or contaminated when prepared or handled by an infected food handler. Pork products may be intrinsically contaminated with HEV and cause a risk of illness if not cooked properly. Since HEV is endemic to many pig populations, control measures focusing on prevention of animal infection on the farm are being proposed.
Viruses differ from bacteria in terms of infectivity, persistence, and epidemiology, posing significant concerns for food safety. Current food safety guidelines often lack validation for foodborne viruses, leading to uncertainties about the effectiveness of control measures. Moreover, existing control strategies are frequently inadequate for managing these viruses, and reliable indicator organisms for viruses are currently unavailable. Good hygienic, agricultural and manufacturing practices are essential to avoid introducing viruses into raw materials and food handling environments, as is the consideration of viruses during the development of HACCP and food safety management plans. Considering that enteric viruses often result in mild symptoms, are difficult or impossible to culture, and that global pathogenic viral surveillance systems are limited, it is expected that the number of foodborne viral disease cases will rise over time as diagnostic methods and surveillance systems improve. In the UK, only one norovirus prevalence study has been conducted to date: the Norovirus Attribution Study performed in 2015/16 (see Further Reading for more details).
Between 1886 and 1898 viruses were first recognised as organisms different from other disease-causing microbes. In 1886, Adolf Mayer was studying tobacco mosaic disease, but tests and attempts to isolate and culture a bacterial agent failed. In 1892, Dmitri Ivanovski demonstrated through an experiment that the disease was either caused by a toxin or by something much smaller than any previously described organism. In 1898, Martinus Beijerinck replicated Ivanovski's experiment and showed that the disease was caused by an infectious life form of some type. This organism became known as tobacco mosaic virus.
In 1914, food was first recognised as a vehicle for viruses when it was identified as the source of an outbreak of raw-milk-associated poliomyelitis. Norovirus was identified in 1968 following an outbreak in a primary school in USA (Norwalk, Ohio), which affected both children and adults. Since the introduction of molecular detection methods in the 1990s, alongside advancements in epidemiology, viruses have been recognised as the leading cause of human gastroenteritis in developed countries.
Viruses do not grow or make their own energy and can only replicate using a living host cell. Outside a host cell they are totally inert, incapable of reproducing or carrying out other metabolic processes and must hijack living cells to replicate. Viruses have, therefore, been considered to lie at, or beyond, the edge of what is considered to be a living organism. Typically, they have an approximately spherical shape with a diameter of 25 to 30 nanometres (nm). They are so small that they can only be observed using an electron microscope. Viruses often show high resistance to stresses typically used in food manufacturing and preservation, such as heat, freezing and UV light. Viruses may be shed in extremely high numbers (up to 107 to 1010 virus particles per gram of stools), and vomit may contain a minimum of 106 particles2. Foodborne viruses have a low infectivity dose of 100 or fewer particles, and this, combined with the high numbers shed, can lead to large outbreaks in a relatively short time.
 |
|
Image courtesy of the Centers for Disease Control and Prevention |
Noroviruses are a highly diverse group of non-enveloped viruses within the Caliciviridae family and are among the leading causes of sporadic infections and epidemic outbreaks of gastroenteritis in humans. The diversity within noroviruses is reflected in their classification into ten genogroups, each containing multiple genotypes. Genogroups GI and GII are primarily responsible for human infections, with GII accounting for a significant portion of norovirus-related illnesses worldwide. Other genogroups (GIII through GX), have host ranges that include pigs, bovines, canines, felines, bats, mice and macaques3. Each genogroup contains several genotypes that exhibit genetic variability. Notably, the GII genogroup includes the most prevalent genotype, GII.4, currently responsible for numerous epidemic outbreaks worldwide. Noroviruses exhibit significant genetic evolution, often undergoing rapid mutations, allowing them to adapt and evade immune responses, contributing to their role as recurrent pathogens. The virus’s single-stranded, positive-sense RNA genome contains three open reading frames (ORFs) encoding various proteins that are crucial for viral replication and assembly. The disease is self-limiting, typically 12-48 hours to 3 days for the majority of people, usually mild, and characterised by nausea, vomiting, diarrhoea, myalgias and abdominal pain. Prolonged virus shedding of up to 8 weeks may occur in asymptomatic people and immunosuppressed individuals. Headache and low-grade fever may occur and there is anecdotal evidence that there may be other diseases caused by norovirus including infant necrotising enterocolitis. The primary route of transmission is person-to-person transmission through the faecal-oral and vomit-oral routes and indirectly through food (ready-to-eat including leafy vegetables and herbs, berries and foods handled after cooking), water and environment. In Europe, outbreaks in healthcare facilities are common.
HAV is a member of the enterovirus group within the Picornaviridae family. Identified in 1973, HAV is one of six hepatitis viruses, designated A, B, C, D, E, and G. Structurally, HAV comprises a single RNA molecule encased within a protein icosahedral capsid measuring 27 to 32nm in diameter and is non-enveloped. The primary mode of transmission is the faecal-oral route. Once ingested, HAV replicates exclusively within liver cells, is excreted in bile, and subsequently shed in the faeces of infected individuals. In developed countries, the incidence of hepatitis A is relatively low. However, outbreaks still occur. For instance, in 2021, an outbreak in the UK was linked to the consumption of Medjool dates. Additionally, frozen berries have been associated with several HAV outbreaks across Europe, emphasising the need for stringent food safety measures.
Hepatitis A is a liver infection which is usually a mild illness characterised by symptoms similar to influenza including sudden onset of fever, malaise, nausea, joint pain, dark-coloured urine, pale stools, anorexia and abdominal discomfort, followed in several days by jaundice, usually with complete recovery within 2 months. The illness can be more severe the older the person is, with infected children under 6 years not experiencing noticeable symptoms. The minimum infectious dose required for HAV infection in humans is unknown but is presumed to be approximately 10 to 100 virus particles, with the incubation period being dependent upon the number of particles ingested (fewer particles mean a longer incubation period). The incubation period for hepatitis A varies from 10 to 50 days (average 28 days) with a long communicability period from early in the incubation period to about a week after the development of jaundice. The middle of the incubation period (2 weeks before symptoms develop) presents the greatest period of communicability, well before the first presentation of symptoms during peak shedding of the virus. Many infections with HAV are asymptomatic, especially in children. Symptoms are occasionally more severe, particularly in people with pre-existing liver conditions with convalescence taking up to 3 months. Patients suffer from feeling chronically tired during convalescence with an inability to work. Fatality is rare, usually occurring in the elderly. Once the virus has been contracted lifelong immunity develops. The UK Health Security Agency publishes updates of Laboratory Reports of HAV infections in England and Wales, the latest covering January to March 2021 (published in January 2024)4.
Routine vaccination of all food handlers is not currently performed, although it could be considered as an additional control measure.
HEV belongs to the genus Orthohepevirus within the family Hepeviridae and has been formally given the taxonomical name Paslahepevirus balayani. It is a positive-stranded RNA virus approximately 7.5 kb in length. The virus is icosahedral in shape, with a diameter of 27−34 nm. The virion is composed entirely of viral protein and RNA. As of 2024, there are eight recognised genotypes (gt) of HEV, each containing several sub genotypes or strains. Genotypes 1 and 2 exclusively infect humans. Genotypes 3 and 4, however, are zoonotic - capable of infecting humans as well as various wild and farmed animals such as pigs, wild boar, rabbits, and deer. Genotypes 5 and 6 are known to infect wild boar. Genotype 7, while primarily associated with camels, has also been recorded in a human infection. Finally, genotype 8 is known to infect camels. The number of confirmed non-travel related hepatitis E cases in the UK has increased particularly since 2010, with similar trends observed in several European countries such as Germany, Netherlands, Spain and the Czech Republic. Several reports have implicated the consumption of specific foodstuffs in the UK as being correlated with HEV infection, but to date, no food has been directly associated with infection in the UK 5, 6, 7.
From the intestinal tract, the virus reaches the liver through an unknown route and mechanism. HEV appears to replicate primarily in liver and gallbladder cells but has been noted in the small intestine, lymph nodes, colon and salivary glands. The incubation period following exposure can range from 3 to 8 weeks, with a mean of 6 weeks. The human infectious dose is currently unknown. The disease is usually mild, asymptomatic and self-resolves in 2 weeks; it is usually seen in age groups 15−40 and can be asymptomatic in children. Symptoms include jaundice, malaise, anorexia, enlarged tender liver, abdominal pain, arthralgia, hepatomegaly, vomiting and fever. Chronic hepatitis has been reported in organ transplant recipients and in patients with active HIV infections. Extended faecal shedding when present occurs for approximately 2 weeks after jaundice develops. Fulminant hepatic failure has been observed especially in pregnant women where mortality rates rise from less than 1% to 25%. In immunocompromised patients, hepatitis E can be persistent and also associated with increased mortality and morbidity in people with progressed liver disease.
Viruses can be considered obligate intracellular parasites because the production of progeny viruses takes place within a host cell. Progeny is formed from the production by the host cell of viral nucleic acid and protein; these viral constituents are capable of ‘self-assembly’ within the cell, spontaneously forming viral progeny. Release of the virus is seldom reliant purely on the lysis of the host cell and can be released in a number of ways.
The viral form that is transmitted from cell to cell and from one host to another is called a particle. If the virus particle’s outer layer contacts a homologous receptor on a susceptible cell’s plasma membrane, the virus’s protein coat, or lipid envelope if present, attaches and infection ensues. To be food or water-borne, a virus must be capable of infectivity upon ingestion by the host in a cell type that is accessible from the digestive tract.
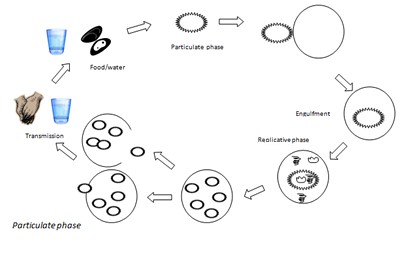
The virus particle attaches and is engulfed by the host cell (usually the whole particle) through what is believed to be a passive process into the cytoplasm – the host cell does all of the work although a high proportion of particles are rejected after coming into contact with a homologous receptor. Uncoating of the protein coat or lipid envelope occurs during the engulfment process with nucleic acid and intrinsic viral enzymes being released into the host cell. As necessary, the viral nucleic acid is transcribed and translated by the cell; this induces production of viral nucleic acid, protein and other constituents required. As quantities are produced, the progeny self-assembly begins; viruses produced in the intestinal tract are called ‘enteric’. If progeny fully mature within the host cell, then particulate release may not be immediate, and the particles may accumulate within the cell; release of the particles is typically gradual rather than the normal bacteriophage ‘burst’. Viruses in animal cells may slowly ‘leak’ through the plasma cell membrane or may stay associated with the cell. The replication cycle may take from between 8 hours to over 24 hours in a single cell.
Enteric viruses are usually resistant to environmental stresses such as heat and acid. The majority are also resistant to freezing and drying, are stable in contact with lipid solvents and may be resistant to ultrahigh hydrostatic pressure. These properties enable foodborne viruses to survive in pickled, marinated and acidic foods. Enteric viruses are capable of surviving and retaining infectivity in marine, estuarine and freshwater for several weeks at 4°C, and survival may be increased by attachment to sediment or particulate matter.
- NoV - noroviruses are resistant to drying and can survive on almost any hard surface (including glass, door handles and railings, for example) for up to 12 hours. Norovirus can survive for at least 56 days on stainless steel and 15 days on carpet. The virus is relatively resistant to high levels of chlorine (up to 10ppm free chlorine) and to varying temperatures. In chilled and frozen environments NoV may be able to survive for months or even years. NoV remains infective after being subjected to pH 2.7 for 3 hours at ambient temperature. While the virus is inactivated by boiling, it can remain infectious at 60°C for 30 minutes and can survive some pasteurisation and steaming processes. It should be noted that since there is no routine and robust infectivity assay yet available for NoV, survival studies have been performed mainly by using surrogate viruses which mimic the characteristics of NoV, or molecular methods (e.g. the reduction of NoV RT-PCR units), so this information is yet to be verified.
- HAV - readily inactivated by heating foods to 85°C for 1 minute but is not subject to thermal denaturation at lower temperatures (70°C for up to 10 min). HAV can survive chilled and frozen temperatures for prolonged periods e.g. several months. Disinfection with a 1:100 dilution of household bleach in water (0.5mg free chlorine for 30 minutes), or cleaning solutions containing quaternary ammonium and/or HCl, are also effective in inactivating HAV, although the virus is resistant to disinfection by some organic solvents and by a pH as low as 3, acid treatment (e.g. pH 1 for 2 h at room temperature) is effective. HAV can survive in the environment for at least 12 weeks at 25°C when excreted in human faeces and can remain infectious after 1 month on environmental surfaces at ambient temperatures, and three to four hours in faecal matter on a person's hand. It has been found to survive for several days in experimentally contaminated freshwater, seawater, wastewater, soils, marine sediment, live oysters, and creme-filled cookies. HAV is more resistant to heat and drying than other enteroviruses.
- HEV - in February 2016, the Food Standards Agency (FSA) and the European Food Safety Authority (EFSA) jointly organised a workshop on foodborne viruses, including HEV. Subsequently, in 2017, EFSA published a scientific opinion on the public health risks associated with HEV as a foodborne pathogen. These findings highlighted significant evidence gaps and identified research priorities, such as developing an assay to evaluate HEV infectivity in food and investigating its thermal stability. Current evidence suggests that heating impacts HEV stability, but there is uncertainty regarding the virus's survival at relevant temperatures and in different food matrices. This complexity is furthered by HEV's ability to exist in various states, with or without a viral envelope, or with a partial envelope, all of which can be present simultaneously in a product. The UK FSA funded additional research, providing further insights. The recommended time/temperature range for short-time cooking is between 65°C and 75°C, but data remain inconsistent. It has been suggested that assays directly evaluating detection or infectivity, such as cell culture or ICC (integrated cell culture)-PCR, are necessary to determine the precise conditions for eliminating HEV8.
It is difficult to estimate the proportion of foodborne disease caused by viruses due to under-reporting, the lack of surveillance systems, often high levels of person-to-person infection, and the inability of existing systems to determine the proportion of disease that is transmitted by foodborne routes relative to other common routes. Increases in population, scarcity of clean water, changes in eating habits such as the increased consumption of food eaten raw, and the globalisation of the supply chain are all contributing to the increase and spread of viral foodborne disease.
The following are just some examples of outbreaks associated with foodborne viruses over the years:
Norovirus
- In May 2005, 2 outbreaks in Denmark were reported associated with the consumption of imported frozen raspberries.
- A cluster of outbreaks was attributed to contaminated commercial ice in 1987 in Wilmington, Delaware.
- In 2005, an outbreak in France was reported associated with the consumption of imported frozen raspberries.
- An outbreak caused by norovirus GII/4 in school lunches, in Tochigi Prefecture, Japan in December 2007 affecting 18 children and 5 adults, possibly linked to salad was reported.
- In January 2009, at a military base in Germany, 36 cases of norovirus were reported, linked to the consumption of contaminated salad.
- January 2010 in Tennessee (USA) 13 people affected by consuming cake in a restaurant.
- February 2012, Missouri (USA), 139 cases associated with fruit salad at a banquet.
- In 2012, the largest recorded outbreak (~12,000 cases) of norovirus in Germany was associated with imported frozen strawberries from China.
- In 2016 in the UK, hundreds of people were ill with norovirus symptoms, which affected both staff and customers, after an outbreak in a popular Mexican restaurant chain.
- In 2017, an outbreak of norovirus at a restaurant chain in the US sickened over 130 people.
- A norovirus outbreak in 2018 in the US and Canada was linked to the consumption of raw oysters from British Columbia, Canada.
- In 2023, norovirus was the suspected cause of a large outbreak at Airbus Atlantic’s Christmas lunch in France, where over 700 staff became ill.
Hepatitis A
- Clam associated outbreak in Shanghai in 1988 with 280,000 people affected due to contamination and inadequate cooking.
- More than 50 residents in South Cambridgeshire (UK) in 1991 where the vehicle was believed to be bread.
- 213 cases in Maine (USA) in 1997 attributed to the consumption of frozen strawberries
- November 2005 in France 111 cases were reported due to the consumption of oysters
- In 2011, seven people were believed to have contracted HAV from sun-dried tomatoes in the UK.
- In 2013, 165 people became ill in a multistate US outbreak attributed to pomegranate arils.
- In 2016, an outbreak in USA was linked to the consumption of frozen strawberries.
- In 2018 six EU countries reported confirmed cases that may have been associated with a single food product.
- In 2021 31 cases in the UK were linked to the consumption of Medjool dates, with more than three-quarters hospitalised.
Hepatitis E has been the cause of sporadic cases and epidemic forms with the primary vehicle being ingestion of faecally contaminated drinking water with foodborne outbreaks involving the consumption of deer meat, wild boar meat, and pork products.
- In New Delhi, in 1955, following the contamination of the city's drinking water, 29,000 cases of icteric hepatitis occurred.
- Indian subcontinent (1975, 1978, 1980) and USSR (1983): several major outbreaks.
- In 2007, in Corsica, seven cases of Hepatitis E were attributed to raw figatellu (a dish made with pig liver).
- In 2024 an outbreak in Finland related to the consumption of pork products resulted in 120 cases, with at least 42 people needing hospital treatment.
Detecting viruses in foods is more difficult than identifying foodborne bacteria. Enteric viruses, such as HAV and NoV, are exceptionally small (approximately 100 times smaller than bacteria) and are usually found in low numbers on foods. Due to their low infectious dose, only minimal quantities of these viruses are needed to pose a health risk when present in food. They do not alter the appearance, taste, or smell of the food, making their presence undetectable through sensory evaluation. Additionally, they cannot proliferate in food, and unlike foodborne bacteria, their numbers cannot be increased through enrichment in nutrient media in the laboratory. Analysts must, therefore, employ complex methods to determine if a food sample is contaminated with viruses. NoV and HAV can be detected in foods using an international standard method (ISO15216 parts 1 & 29, 10). This method is suitable for use on shellfish, fresh and frozen produce such as leafy greens and soft berry fruits, bottled water and food contact surfaces. Testing is based on the detection of viral nucleic acids by real-time PCR and therefore does not provide any indication of infectivity. Although there are several methods to detect HEV already published11, there is currently no standardised method available; efforts to develop such a method are underway by the working group ISO/TC 34/SC 9/WG 31 within the International Organization for Standardization (ISO). Although detection of foodborne viruses has been performed for many years, it is still not considered a routine method, due to the lack of regulation or criteria for limits of viruses in foods. In the UK there are very few laboratories with either the expertise or capability to detect viruses in foods.
Good Industry Practice
Controls to reduce the risk of foodborne transmission of foodborne enteric viruses should focus on 12, 13, 14, 15:
- Use of potable water for irrigation and processing.
- Use of shellfish from approved waters.
- Active monitoring of shellfish production areas for contamination events.
- Thorough cooking of shellfish prior to consumption (85−90°C for 4 minutes or steamed for 90 seconds).
- Excluding employees suffering from gastroenteritis for 48 hours after symptoms have ceased and 1 to 2 weeks after the onset of jaundice.
- Exclude symptomatic food handlers from the entire food business site (not just food handling duties and areas). Once excluded, they should remain away for a further 48 hours from when symptoms stop or from the end of any treatment of the symptoms with medicine such as anti-diarrhoeal drugs.
- Food handlers with Hepatitis A should remain off work for seven days after the onset of jaundice and/or other symptoms. Any food handler who develops jaundice for an unknown reason should be excluded immediately and seek medical advice.
- Effective training in personal hygiene; practical advice about hand washing techniques and education on when to seek medical attention.
- Thorough cleaning with an effective disinfectant following any vomiting episode in a handling environment.
- Destroying any food that may have become contaminated through aerosols and cleaning up and disinfecting thoroughly over a wide area after someone has been sick in or near a food handling area,
- Easy access to hand washing and sanitary facilities for all employees (including field workers).
- Discourage children from areas where food is harvested, handled or processed.
- Correct disposal procedures for sanitary waste.
- Potable water or chlorinated water should be used for rinsing produce or for making ice for packing or consumption.
- Posting handwashing signs, for after eating and visiting the toilet, with frequent written and verbal reminders.
- Development of a HACCP including the risks and control measures for foodborne viruses.
Specific guidelines already exist for NoV and HAV which have been published by Codex Alimentarius16. Revised guidelines are currently in progress (see https://food.ec.europa.eu/document/download/ba016f74-25f7-4643-a39e-5fd8adbec224_en?filename=codex_ccfh_54_agenda-item_10.pdf) and it is anticipated that HEV will be included in these revised guidelines.
- Summary report of the Joint FAO/WHO Expert Meeting on the Microbiological Risk Assessment of Viruses in Foods Part 1: Food Attribution, Analytical Methods, and Indicators. 2023.
- Miller, C., Fraser, A., Sturgis, R., Saunders, A., & Chen, X (2013). Public Health Reasons: Vomiting and Faecal Episodes. Department of Food, Nutrition, and Packaging Sciences, Clemson University, Clemson, SC 29634.
- Chhabra, P., de Graaf, M., Parra, G.I., Chan, M.C.-W., Green, K., Martella, V., Wang, Q., White, P.A., Katayama, K., Vennema, H., Koopmans, M.P.G., & Vinjé, J. (2019). Updated classification of norovirus genogroups and genotypes. Journal of General Virology, 100, 1393–1406. https://doi.org/10.1099/jgv.0.001318
- UK Government. (2024). Laboratory Reports of Hepatitis A Infections in England and Wales: January to March 2021. https://www.gov.uk/government/publications/hepatitis-a-england-and-wales-2021/laboratory-reports-of-hepatitis-a-infections-in-england-and-wales-january-to-march-2021
- Said, B., Ijaz, S., Kafatos, G., Booth, L., Thomas, H.L., Walsh, A., Ramsay, M., & Morgan, D. (2009). Hepatitis E outbreak on cruise ship. Hepatitis E Incident Investigation Team. Emerging Infectious Diseases, 15(11), 1738-1744. https://doi.org/10.3201/eid1511.091094
- Said, B., Ijaz, S., Chand, M.A., Kafatos, G., Tedder, R., & Morgan, D. (2014). Hepatitis E virus in England and Wales: Indigenous infection is associated with the consumption of processed pork products. Epidemiology and Infection, 142(7), 1467-1475. https://doi.org/10.1017/S0950268813002318
- Said, B., Usdin, M., Warburton, F., Ijaz, S., Tedder, R.S., & Morgan, D. (2017). Pork products associated with human infection caused by an emerging phylotype of hepatitis E virus in England and Wales. Epidemiology and Infection, 145(12), 2417-2423. https://doi.org/10.1017/S0950268817001388
- Food Standards Agency. Thermal Inactivation Model for Hepatitis E Virus (HEV). https://www.food.gov.uk/research/foodborne-disease/thermal-inactivation-model-for-hepatitis-e-virus-hev
- Anonymous. (2017). ISO 15216-1:2017 Microbiology of the Food Chain – Horizontal Method for Determination of Hepatitis A Virus and Norovirus using Real-time RT-PCR — Part 1: Method for Quantification. International Standards Organisation, Geneva.
- Anonymous. (2019). ISO 15216-2:2019 Microbiology of the Food Chain — Horizontal Method for Determination of Hepatitis A Virus and Norovirus using Real-time RT-PCR — Part 2: Method for Detection. International Standards Organisation, Geneva.
- Cook, N., D’Agostino, M., Wood, A., & Scobie, L. (2022). Real-time PCR-based methods for detection of hepatitis E virus in pork products: A critical review. Microorganisms, 10, 428.
- Todd, E., & Grieg, J. (2015). Viruses of foodborne origin: A review. Virus Adaptation and Treatment, 7, 25-45.
- Bosch, A., Gkogka, E., Le Guyader, F.S., Loisy-Hamon, F., Lee, A., van Lieshout, L., Marthi, B., Myrmel, M., Sansom, A., Schultz, A.C., Winkler, A., Zuber, S., & Phister, T. (2018). Foodborne viruses: Detection, risk assessment, and control options in food processing. International Journal of Food Microbiology, 285, 110-112. https://doi.org/10.1016/j.ijfoodmicro.2018.06.001
- Chilled Food Association. (2016). Microbiological Guidance for Produce Suppliers to Chilled Food Manufacturers (3rd ed.).
- Food Standards Agency. (2009). Food Handlers Fitness to Work. Regulatory Guidance and Best Practice Advice for Food Business Operators. https://www.food.gov.uk/sites/default/files/media/document/fitnesstowork...
- Codex Alimentarius (2012). Guidelines on the application of general principles of food hygiene to the control of viruses in food
https://www.fao.org/fao-who-codexalimentarius/sh-proxy/en/?lnk=1&url=htt...
Further reading
- Koopmans M, Duizer E (2004). Foodborne viruses: an emerging problem. International Journal of Food Microbiology 90 (1), 23–41.
- Oogane T, Hirata A, Funatogawa K, Kobayashi K, Sato T, Kimura H (2008). Food poisoning outbreak caused by norovirus GII/4 in school lunch, Tochigi prefecture, Japan. Jpn J Infect Dis 61 (5), 423−424.
- Westrell T, Dusch V, Ethelberg S, Harris J, Hjertqvist M, Jourdan-da Silva N, Koller A, Lenglet A, Lisby M, Vold L (2010). Norovirus outbreaks linked to oyster consumption in the United Kingdom, Norway, France, Sweden and Denmark, 2010. Euro Surveill 15 (12), pii=19524. Available from: http://www.eurosurveillance.org/ViewArticle.aspx?ArticleId=19524
- Korsager B, Hede S, Bøggild H, Böttiger BE, Mølbak K (2005). Two outbreaks of norovirus infections associated with the consumption of imported frozen raspberries, Denmark, May−June 2005. Euro Surveill 10(6), E050623.1.
- Wadl M, Scherer K, Nielsen S, Diedrich S, Ellerbroek L, Frank C, Gatzer R, Hoehne M, Johne R, Klein G, Koch J, Schulenburg J, Thielbein U, Stark K, Bernard H (2010). Food-borne norovirus-outbreak at a military base, Germany, 2009. BMC Infect Dis 10, 30.
- Bernard H, Faber M, Wilking H, Haller S, Höhle M, Schielke A, Ducomble T, Siffczyk C, Merbecks S S, Fricke G, Hamouda O, Stark K, Werber D, on behalf of the Outbreak Investigation Team. Large multistate outbreak of norovirus gastroenteritis associated with frozen strawberries, Germany, 2012. Euro Surveill. 2014;19(8):pii=20719. https://doi.org/10.2807/1560-7917.ES2014.19.8.20719
- Morgan Mari, Watts Vicky, Allen David, Curtis Daniele, Kirolos Amir, Macdonald Neil, Maslen Ellie, Morgan Deb, Saei Ayoub, Sedgwick James, Stevenson Janet, Turbitt Deborah, Vivancos Roberto, Waugh Catriona, Williams Chris, Decraene Valerie. Challenges of investigating a large food-borne norovirus outbreak across all branches of a restaurant group in the United Kingdom, October 2016. Euro Surveill. 2019;24(18):pii=1800511. https://doi.org/10.2807/1560-7917.ES.2019.24.18.1800511
- Norovirus Attribution Study: Assessing the contribution made by the food chain to the burden of UK-acquired norovirus infection. https://www.food.gov.uk/research/foodborne-disease/norovirus-attribution...
- Srinivasan Mani, Snehashis Hazra, Joseph Hagan, Amy Sisson, Jayasree Nair, Mohan Pammi; Viral Infections and Neonatal Necrotizing Enterocolitis: A Meta-analysis. Pediatrics July 2023; 152 (1): e2022060876. 10.1542/peds.2022-060876
- Food Standards Agency. Food Handlers: Fitness to Work Regulatory Guidance and Best Practice Advice For Food Business. Available from: https://www.food.gov.uk/sites/default/files/media/document/personal-hygi...
- Hassard, F., Sharp, J.H., Taft, H. et al. Critical Review on the Public Health Impact of Norovirus Contamination in Shellfish and the Environment: A UK Perspective. Food Environ Virol 9, 123–141 (2017). https://doi.org/10.1007/s12560-017-9279-3
Institute of Food Science & Technology has authorised the publication of the following updated Information Statement on Foodborne Viruses, dated September 2024, replacing that of September 2018. This updated Information Statement has been prepared and peer reviewed by professional members of IFST and approved by the IFST Scientific Committee.
The Institute takes every possible care in compiling, preparing and issuing the information contained in IFST Information Statements, but can accept no liability whatsoever in connection with them. Nothing in them should be construed as absolving anyone from complying with legal requirements. They are provided for general information and guidance and to express expert professional interpretation and opinion, on important food-related issues.
