August 2022
Introduction
Food allergy, coeliac condition, and food intolerance are aspects of food hypersensitivity.
Briefly, in susceptible individuals:
- food allergy is an acute illness caused by proteins in food that interact with the immune system via allergen-specific immunoglobulin E, IgE;
- coeliac condition is a chronic illness caused by gluten, a collection of proteins in wheat and certain other cereals; and
- food intolerance is a chronic reaction to one or more of a range of chemical compounds in food, such as lactose (lactose intolerance), histamine (scombrotoxic food poisoning), and FODMAPS[1] (short-chain carbohydrates that may cause digestive symptoms including gas, bloating, pain, diarrhoea, or constipation);
- consumers may also choose to avoid certain foods. This may be due to a religious or cultural diet, or preference e.g. for plant-based foods.
Many, but not all, foods causing hypersensitivity are covered by labelling obligations in most jurisdictions. The EU and UK maintain one of the most extensive lists, in Annex II of Regulation 1169/2011 on food information to consumers[2]. This resource deals mainly with sampling and analysis of the food groups listed in Annex II and discusses other common analytical targets omitted from Annex II. Updates on international regulation of priority allergens in different jurisdictions are available from the University of Nebraska Food Allergy Research and Resource Program (FARRP) Food Allergens - International Regulatory Chart.
See IFST Information Statement: 'Food Allergy' for more information.
[1] FODMAPS: Fermentable Oligosaccharides, Disaccharides, Monosaccharides and Polyols
Analysis may be required for a variety of reasons many of which are based on the needs of some form of risk assessment:
- Protect consumers at risk
- Establish allergen status of raw materials/ingredients
- Identify equipment that is difficult to clean
- Validate the effectiveness of cleaning, swabbing/allergen changeovers
- Verify the risk of allergen carry-over in the finished product
- Confirm free-from claims
- Monitor the effect of critical changes
- Inform the risk assessment in incidents and complaints
- Enforcement and forensic: seized exhibits, replica meals[1]
Having identified the purpose, we need to think about sampling and how the sample is handled, what type of analysis we will carry out and what method we will apply. We need to consider how to gain assurance that the method is fit for purpose, and how the results are reported. The results must be interpreted in the context of the risk assessment. Figure 1 summarises these steps.
Figure 1: Analytical route map (Practical Guidance on the Application of Allergen Quantitative Risk Assessment - Courtesy of ILSI-Europe, 2022)
[1] Replica meals are made up by a catering outlet to mimic the meal suspected of having caused an allergic reaction, presumably by cross-contamination, when the original meal or its remnants are unavailable for analysis. Usually requested by an Environmental Health officer (EHO), Trading Standards Officer (TSO), or the police investigating a serious allergic reaction.
Owing to the complexity of allergen analysis it is essential that customers of analytical services and their laboratories enter into dialogue to establish their mutual needs. Results that seem ‘unexpected’ should especially be discussed. Bear in mind that any subsequent laboratory investigations may range from the simple to the complex, with time and budget allocated accordingly, examples below:
| Examples of laboratory investigations | Complexity |
| First report was a ‘greater than’ figure, as the signal was above the highest point on the calibration curve. Sample must be diluted and re-run |
Simple Complex |
| Result is sufficiently concerning with potentially significant consequences to be re-run by a second technique (a different ELISA, qPCR or LC-MS) | |
| Matrix suppression or enhancement suspected, further dilutions needed and re-run, spike and recovery experiments, or re-run with a second ELISA | |
| Previously unreported method cross-reactivity, investigated by application of complementary techniques |
Analytical results are only useful if the sample submitted to the laboratory was meaningfully taken. See IFST Information Statement: 'Sampling for Food Analysis'. However, there is relatively little practical guidance on sampling for analysis related to food hypersensitivity. To fill this gap, guidance has been produced by the European section of the International Life Sciences Institute (ILSI-Europe). A brief overview is presented here. Sampling and analysis of raw materials to verify they are what they are claimed to be is relatively straightforward, although not trivial and not dealt with here. The more complex question is often “is there any unintended allergen present and if so, at what concentration?”. Figure 2 illustrates a strategy to determine the number of samples required.
Figure 2: Strategy to determine the appropriate number of samples
The ILSI guidance envisages three levels of concern, low medium and high. The sorts of common and different factors bearing on these are shown in figure 2. At a low level of concern, if present at all, the unintended allergen is known to turn up regularly and homogenously in the food product of interest. A small number of samples will usually suffice to give useful quantitative analytical data. As concern increases, less can be assumed about the frequency and homogeneity of the unintended allergen presence. If it is likely to be particulate in nature, irregularly and unpredictably distributed in the food product greater numbers of samples are required to characterise both the amount present and the dispersion of the data. Here, rules of thumb provide much-needed consistency to the sampling plan. One such is to take the cubed root of the number of units in the consignment as the number of samples to be taken and individually analysed. In particularly challenging circumstances the sampling plan should involve a much larger number of samples, e.g. the square root of the number of units plus one. This becomes akin to the sort of sampling plan necessary for mycotoxin analysis, and for the same reason, ‘hot-spots’ in the consignment. There are other things to think about such as increasing sample and test portion sizes to reduce dispersion. Please see the ILSI-Europe guidance and references therein.
The purpose of analysis often dictates the type of test applied. Food intolerance compounds, other than FODMAPS, are mainly small molecules and a variety of techniques are applied to their analysis. We will touch on some of these later.
Concentrating on allergens, we may want rapidly to check incoming raw materials, using lateral flow devices. For validation and verification of cleaning to prevent cross-contamination, swabbing of surfaces is deployed which may be analysed on-site or sent to the laboratory. For the other purposes, we will touch on the main techniques, complementary methods, and orthogonal confirmation. Figure 3 shows an overview of allergen analysis.
Figure 3: Overview of allergen analysis
Reprinted from Encyclopedia of Food Chemistry, 1st Edition, Walker, M.J., Food Allergens: An Update on Analytical Methods, Pages 622-639., Copyright (2022), with permission from Elsevier. License number: 5344130466784
There are many techniques for allergen analysis. From left to right across figure 3, lateral flow devices (LFD), surface swabs or rinse water analyses are carried out by some form of ELISA. Personal allergen testing is a developing field using hand-held devices often connected to a smartphone, but that’s a subject for another day.
Routine laboratory analysis is still mainly by ELISA, although PCR and LC-MS are also available, while biosensors are an area of growing interest for allergen analysis, although time precludes us from going into detail. Research proteomics is a specialised field that we won’t touch on in this IFST Information Statement. Allergen analysis by any technique is relatively more expensive than, for example, a basic microbiological test suite, because of the current complexity of such work.
Glossary of analytical terms:
- ELISA: Enzyme Linked ImmunoSorbent Assay targeting proteins
- LFD: Lateral Flow Device, a rapid test based on ELISA
- Real Time PCR (qPCR): Polymerase Chain Reaction, targeting DNA
- HPLC: High Performance Liquid Chromatography
- LC-MS: Liquid Chromatography-Mass Spectrometry, including LC-MS/MS, tandem Mass Spectrometry
- SDS-PAGE: Sodium Dodecyl Sulphate-Polyacrylamide Gel Electrophoresis
- ToF MS: Time-of-Flight Mass Spectrometry
Figure 4 gives a summary of the main analytical techniques applied to food hypersensitivity analyses:
|
Test method |
Image |
Comments |
Advantages |
Disadvantages |
Typical uses |
|
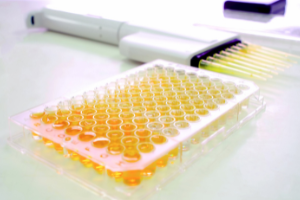
Enzyme Linked ImmunoSorbent Assay ELISA |
|
Targeting proteins |
Capable of standardisation, quantified results and good precision. Relatively affordable if operated batch wise |
Results can be kit |
All priority allergens except celery |
|
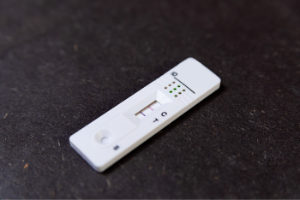
Lateral Flow Device LFD |
|
Rapid test based on ELISA Targeting proteins |
Offer low cost rapid simple point of use assays capable of use by nontechnical staff |
Exhibiting similar specificity and limits of detection to ELISAs, they are semiquantitative with subjective quantitation and reproducibility can be less than with ELISA |
Most priority allergens except celery, uses include checking incoming raw materials |
|
Polymerase Chain Reaction, Real Time PCR qPCR |
|
Targeting DNA |
Quantitative results can be obtained. Valuable for foods for which no ELISA based test is available, relatively affordable |
Do not directly measure the hazard (protein). Requires strict physical separation in the laboratory of extraction and amplification |
Most priority allergen species except milk and egg |
|
High Performance Liquid Chromatography HPLC |
|
Targets small molecule analytes |
Ubiquitous versatile laboratory systems, relatively inexpensive |
Mainly small molecule analytes rather than large protein molecules |
Examples include: Histamine, Annatto, Tartrazine, Sulphites, Benzoic acid |
|
Liquid Chromatography – Mass Spectrometry (including LC-MS/MS, tandem M/S) LC-MS LC-MS/MS |
Courtesy of IGFS QUB, © QUB |
Targeting proteins via peptides. Extracting the allergenic proteins from a food matrix, followed by enzymatic digestion e.g. with trypsin |
Mass spectrometry allows harsher extraction conditions than can be used with ELISA. The peptides can then be chromatographed and detected as with small molecule MS. Multiplexing automated runs are the norm and metrological traceability is achievable |
Expensive to set up and run. |
Most priority allergen |
|
Biosensors |
|
Targets analyte (protein)
Optical biosensors and Surface Plasmon Resonance (SPR) are most common |
Direct, reproducible, real-time and relatively inexpensive Nanosensor applications have low detection limits (single molecule detection), reduction of reagent volumes, shorter analysis times or multianalyte detection, improved sensitivity, selectivity, robustness, accuracy, higher efficiency and sample throughput in the analysis of complex food samples
Can be used to detect protein or DNA
|
Expensive set up costs
Relatively few sensors based on nanoparticles have been described, for the analysis of food allergens
New methods are required, for the detection of trace concentrations of allergens in complex food matrices, as well as for hidden food allergens
|
As yet, most applications remain in research or in clinical settings |
Figure 4: Summary of the main analytical techniques applied to food hypersensitivity analyses.
Food allergens and gluten are medium to large protein molecules and without ELISA it would, for a long time, have been impossible to detect and determine food allergens. ELISA makes use of antibodies, generally raised in animal hosts, to bind with high selectivity to antigens, in this case the allergenic proteins. One way to look at it is that the technology resides in the antibodies rather than the apparatus (Allen, 1986). ELISA is the current workhorse technique for allergens. Its immunological basis makes it quite specific to the target allergenic foods, but we need to bear in mind that different ELISA platforms often give different quantitative results. Why is this so? Different kits may have differing extraction efficiencies, which is also affected by the nature of the sample matrix. The antibodies essential for the detection of proteins are often raised against different targets for the same allergenic food, and if different protein isoforms affected by differing post translational modification occur in the sample, differing antibody binding efficiency will occur. The processing history of the food may also disrupt the protein structure sufficiently to result in reduced antibody binding. These factors give rise to differing quantitative data from various ELISAs, as seen in multi-modal datasets in proficiency test rounds for allergens analysis. Cross-reactivity may also be a problem although ELISA kit manufacturers, in each method validation, check against over 30 potential foods that may cross react. It is quantitative, easy to use in a laboratory without expensive equipment, and quite rapid, although at least a day is required for an analytical run and it makes sense to batch samples for the same analytical target. ELISA exhibits suitable limits of detection for many allergens. With certain caveats, ELISA is relevant and largely accepted by the food industry.
Figure 5 is a simplified explanation of the ELISA workflow.
Figure 5: ELISA workflow.
Step 1 shows the capture antibody which is specific to the intended allergen preloaded into the microtitre plate. In step 2, an extract of the sample in a suitable buffer is added to the well and any allergen present binds to the antibody. A second antibody, to which an enzyme has been bound is introduced and binds to the allergen. After washing the microtitre plate free of reagents a colourless substrate is added on which the enzyme acts. This produces a colour in the well, the absorbance of which is proportional to the amount of allergen captured and can be measured via spectrophotometry in a plate reader.
The protein targeted by the ELISA must be relevant. For example, a milk ELISA that determines ß-lactoglobulin (BLG) will not be appropriate if it is necessary to determine casein.
Cross-reactivity is defined as a positive response to a sample that does not contain any of the target analyte (Abbott et al., 2010). Those who develop allergen ELISAs routinely check their assay for cross-reactivity against a wide battery of food commodities. Particular care is needed with foods that are genetically similar to the target because of the higher probability of expression of proteins with amino acid sequence homologies. Examples include:
- Cross-reactivity between members of the Prunus family including apricot, cherry, peach, plum and almond, as well as mahaleb (Prunus mahaleb L.)
- Cross-reactivity between peanut and soya, soya is closely related to other members of the Leguminosae family as shown by Lacorn et al., 2018.
- The most well-known example of ELISA cross-reactivity is within the Apiaceae family, also known as the Umbelliferae, or, more familiarly, as the celery, carrot, or parsley family. Owing to extensive cross-reactivity, a specific ELISA is not possible for celery, which necessitates a PCR approach.
- A relatively recent emerging cross-reactivity is with regard to several mustard ELISAs which also react to canola protein. This can be solved by PCR provided the primers/probes have been validated as negative to the relevant Brassica species. A further problem has arisen with cross-reactivity to Sinapis arvensis, the wild mustard or charlock, for which PCR is also cross-reactive. An LC-MS method remains to be validated and this issue is a current problem.
- Clinical experience of patients cross-reacting to different foods that possess similar proteins is often a guide to laboratory cross-reactivity. For example, Lipid Transfer Proteins (LTPs) are found in plants and foods that contain plants. Lipid Transfer Protein Syndrome is an allergy affecting people who have become sensitised to LTPs. LTPs are small proteins found in a wide variety of plant foods; common foods include tomatoes, apples, raspberries, stoned fruit, grapes, walnuts, almonds, corn, and barley. Patients may react to one or multiple foods.
For commodities such as crustaceans and fish, where multi-species allergies are common, cross-reactivity may be welcomed as a screening approach.
Reference materials (RMs) contain known amounts of the target analyte. They are widely essential for the proper validation and quality control of analytical methods and are particularly called for in allergen analysis. RMs and Certified Reference Materials (CRMs) are established concepts with a well-defined vocabulary defined by ISO 17034:2016 ‘General requirements for the competence of reference material producers’ with reference materials produced by National Measurement Institutes or by Designated Institutes exhibiting the highest standards.
RM producers must ensure sufficient material homogeneity and stability, assess the incurred quantity and, for matrix allergen materials, RMs should be industrially realistic for processed food. The incurred concentrations should be appropriate and establish a relationship with concentrations that affect those with allergies. Making an RM is, therefore, a complex process, relatively expensive, seldom a commercial proposition, and presents particular practical difficulties with regard to allergens. There are few RMs produced solely with allergen analysis in mind.
CRMs require a reference method traceable to the International System of Units (SI) to be available to characterise the incurred amount (the ‘measurand’). As of mid-2022, only one such reference method has been published, see Breidbach et al., 2022, from which a milk CRM may be expected in the coming years.
The uses of incurred food allergen RMs include:
- validation and verification studies of analytical methods (ELISA, qPCR, LC-MS/MS, LFD), and relevant ELISA and qPCR kits
- validation studies of ELISA, qPCR and LFD kits
- method and kit development
- competency assessments of analytical service providers and staff
- the incurred RMs can also be used to optimise analytical recovery from similar matrix types to inform risk assessors of the possible ‘true’ estimate of allergen in a questioned product.
Raw material RMs can be used to generate kit calibrator extract solutions and generate external check calibrator extract solutions. It will be impossible to make RMs for the required wide variety of matrices that are analysed by laboratories (e.g., takeaway curry-type products); however, raw material RMs may be used to prepare fortified (‘spiked’) matrices either by way of an extract or, preferably, by addition of the raw material itself to assess recovery in real life situations.
A blank matrix can be used as a ‘no-template’ control to provide assurance of the absence of in-laboratory allergen cross-contamination (either environmentally, from personnel, or in reagents), and calculate the method Limit of Detection or Limit of Quantification (LoD or LoQ).
RMs, and especially CRMs when they become available, are not necessarily intended to be used habitually in routine analytical runs. A more economical approach is to use the RM to validate in-house quality control materials. The latter should of course be checked for homogeneity and stability. It is also possible to prepare in-house quality control materials when RMs are not available for the allergen/matrix combination being analysed.
Available RMs for allergen analysis LGC Standards, made by the National Measurement Laboratory (NML) the International Association for Monitoring and Quality Assurance in the Total Food Supply Chain (MoniQA), and The National Institute of Standards and Technology (NIST) are shown below, although other examples are available Standards Light roasted partially defatted peanut flour (LGCQC1020).
- Chocolate dessert matrix: Incurred, 10 mg/kg as peanut protein, & a negative control (< 1mg/kg as protein).
- Allergen kit: milk, egg white, almond, hazelnut, and walnut (LGC746-KT) raw material RMs & incurred, at 10, 10, 9.7, 9.8 & 10 mg/kg as protein respectively in a chocolate matrix and a separate chocolate matric ‘blank’ for milk, egg white, and hazelnut proteins. Produced under an LGC-led project, funded by the Food Standards Agency (FSA) with contributions from the University of Manchester and Romer Laboratories Ltd.]
MoniQA
- Set of materials for milk allergen analysis which include dried skimmed milk powder, blank matrix (gluten-free cookie), and two incurred materials.
NIST
A note of caution, however, was sounded in a paper by Ryzchon et al. (2017) on comparisons between paired data from seven gluten ELISAs with various calibrators. Strong evidence of bias or outliers in the paired results confirmed a lack of correlation, suggesting the kits target different gluten markers, the markers may not be present in gluten at a constant ratio, and/or food processing may alter the markers.
RMs will not necessarily improve measurements where this lack of correlation is observed, although their use will highlight the variation which will be of interest to risk assessors.
With the correlation between test kits, the application of a reference material improved the comparability of results. But the beneficial effects are not observed equally by all kits or even for all matrices using the same kit.
The Polymerase Chain Reaction deployed in real-time PCR (qPCR) is readily available to detect and quantify the amount of allergen species-specific DNA in a sample.
For some foods, for example, celery, no ELISA is available owing to cross-reactivity in the botanical family to which celery belongs. Celery is thus routinely assayed by qPCR.
The advantages of qPCR include that DNA is often a more robust molecule than many proteins. qPCR gives the required sensitivity, multiplexing, and DNA specificity in a relatively rapid, uncomplicated setup and workflow. There are many real-time PCR instruments available and the application of qPCR gives complementarity when confirmation of the species is required.
The main disadvantage leveled against qPCR is that the analytical target is DNA rather than protein, which is the hazard. This is valid, and for egg and milk, DNA methods are not applicable since it is not possible to distinguish DNA from hens’ eggs from DNA from chicken meat. Similarly, cows’ milk and beef both contain bovine DNA. However, in most other cases validated qPCR findings should not be dismissed without detailed evidence that no protein is present.
Quantification of the concentration of unintended allergen species present in a sample is possible, initially on a DNA copy number to copy number basis, unless a suitable food-based reference material (RM) is used to construct the calibration curve. More meaningful quantification requires conversion factors, that remain to be fully harmonised. Digital PCR (dPCR) is capable of absolute quantification without a calibrator.
For more information, refer to the AMC Technical Brief on PCR.
Liquid chromatography coupled to mass spectrometry, more usually tandem mass spectrometry, LC-MS/MS, is the ‘gold standard’ for small molecule target identification and quantification. It is routinely applied to food allergen proteins, often as a multiplexed method to detect several allergens in the same analytical run. Extracted protein is reproducibly digested using enzymes such as trypsin. The resulting peptides, often 6 to 12 amino acids in length and with a double or triple charge bring the mass to charge ratio well within the range of typical small molecule mass-to-charge (m/z) making LC-MS/MS eminently feasible. Provided at least two peptides uniquely representing the protein target are demonstrably available, identification of the protein is assured. Moreover, LC-MS/MS is the only technique that has been shown to be capable of providing traceability to the SI, bringing with it the ability to harmonise allergen protein measurements on a global basis. However, the technique is costly, requiring high-end instrumentation, highly skilled operators and, ideally, isotopically labelled internal standard peptides for quantification. The workflow is sophisticated and can be time consuming. This may explain why routine allergen quantification by LC-MS/MS is as yet not routinely practiced or offered by many analytical service laboratories. However, for critical problems it may be the best approach.
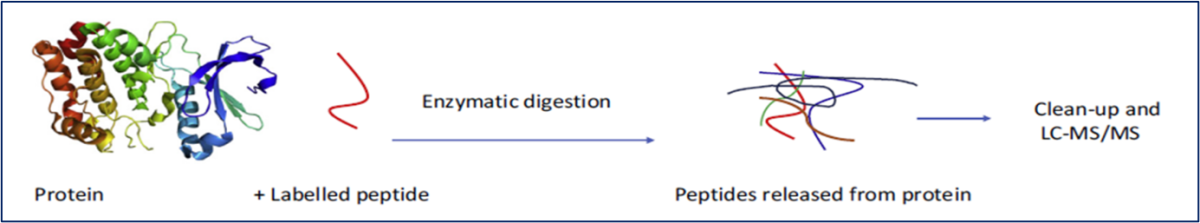
Figure 6: LC-MS/MS workflow
Biosensors are proposed as potential alternatives to traditional methods, such as ELISA, for direct, reproducible, real-time, and relatively inexpensive detection of food allergens. They are analytical devices incorporating a biological, or biologically derived, sensing element (such as antibodies), either intimately associated with, or integrated within a physicochemical transducer. The transducer converts the recognition event into a measurable chemical or physical signal, to produce a digital electronic signal proportional to the concentration of a specific analyte, or group of analytes. These can be generic proteins, from the food causing the allergenic effect, or specific biomarker proteins identified for different food allergens.
The combination of new selective bio- and biomimetic receptors, with the unique optical, electrical, or electrochemical properties of nanomaterials, has allowed the development of new nanosensors with the potential for improvements in food safety, quality control, and traceability. They offer excellent prospects for the development of portable, miniaturised, real-time, easy-to-use, rapid, and cost-effective sensors capable of screening multiple allergens when required. Some of the advantages of nanosensor applications, for allergen detection, include low detection limits (single molecule detection), reduction of reagent volumes, shorter analysis times, or multianalyte detection. Their use has also led to improved sensitivity, selectivity, robustness, and accuracy of conventional analytical methods, higher efficiency and sample throughput, as well as better performance for the analysis of complex food samples. Relatively few sensors based on nanoparticles have been described for the analysis of food allergens.
From an analytical perspective, new methods are required for the detection of trace concentrations of allergens in complex food matrices, as well as for hidden food allergens, or new allergenic foods prior to them being commercially available. Other challenges in this field include the exploration of new ways to enhance the signal-to-noise ratio, signal amplification, enhancement of the transduction efficiency, sample preparation, and biosensor validation. In any case, it is vital to connect laboratory research with real-world applications. Biosensors are of interest to the food industry and regulatory bodies, responsible for the protection of the health of food-allergic consumers. The food industry could play an essential role by encouraging research in this field. At the time of writing biosensor applications for food allergen analysis largely remain in the research and clinical fields.
A good example of complementary application of ELISA, PCR, and LC-MS/MS was the solution to the problem of alleged contamination of cumin and paprika by almond, an Annex II allergen. Following suspicions that the initial finding of almond (Prunus dulcis) in cumin was due to ELISA cross-reactivity to a related species Prunus mahaleb, the cross-reactivity was confirmed. In the absence of any specific assay for mahaleb, a combination of ELISA, PCR, and LC-MS/MS was required to show that the cumin was contaminated by mahaleb and the paprika by almond. The full story can be read in the open access paper ‘Almond or Mahaleb? Resolution of allergen ELISA findings in cumin and paprika by molecular biology and protein mass spectrometry’.
In highly critical situations, complementary approaches to provide either weight of evidence or orthogonal confirmation may be required. For example:
- two different ELISA platforms, preferably offering different detection antibodies, and calibrators; or
- ELISA plus PCR; or
- ELISA or PCR + LC-MS/MS; or
- all three may be needed.
This advice can be difficult for analytical service labs to apply since they have put a lot of effort and funding into gaining ISO/IEC 17025 accreditation for a specific assay. Few if any labs can justify the cost of parallel accreditation by a different platform. However, a flexible scope may be of assistance; even checking with a lateral flow device built using different antibodies can yield useful information. Sub-contracting to another laboratory with a different accredited assay would be better than reporting a result that subsequently turns out to be wrong owing to cross-reactivity.
It is axiomatic that laboratories carrying out food hypersensitivity related analysis have quality assurance in place - planned and systematic activities within the quality system. These include familiarisation with and validation of the method. Validation includes assessment of the method performance characteristics, typically accuracy (trueness, or ‘bias’), applicability (for the matrix and concentration range), the limit of detection, limit of quantification, precision (repeatability including intermediate precision and reproducibility), selectivity, sensitivity, linearity and uncertainty. These terms are simply explained in the IFST Information Statement: ‘How to Choose and Instruct a Laboratory for Chemical Food Analysis’ which references the authoritative international texts on the subject. Quality control activities to monitor the performance of the method, e.g. control charts, and accreditation to ISO/IEC 17025 ‘General requirements for the competence of testing and calibration laboratories’, provide assurance that the method remains fit for purpose.
For ELISA, special focus should also be on checking for cross reactivity, matrix interference, and practical issues such as pipetting, plate washing, calibration curves and the use of in-house and external RMs and proficiency testing rounds. It is also important to remember that the reactions in an ELISA may not have time to come to equilibrium if the assay is to be completed within the time allocated hence the kit manufacturer’s procedure must be followed exactly, especially as to timing and temperatures.
In qPCR the same considerations of quality assurance and quality control apply and in addition special attention is required to the quantity and quality of the extracted DNA. Measurement of DNA concentration and purity can be by optical density (absorbance), agarose gel electrophoresis or fluorescent DNA-intercalating dyes. Absorbance at 260 nm can be used to calculate the concentration of nucleic acids in solution and for reliable spectrophotometric DNA quantification, a series of dilutions is recommended. Strong absorbance close to 230 nm indicates organic compounds or chaotropic salts in the extracted DNA. Substances including urea, EDTA, guanidine hydrochloride, and phenolate ions all exhibit absorbance maxima at or close to 230 nm. The absorbance ratio at 260 nm over 230 nm should be greater than 1.5, and preferably close to 1.8. Values that differ significantly from these target values are usually indicative of extraction reagent carryover. See also the specific advice on quantification by qPCR by Burns (2019).
Again, for LC-MS/MS the same considerations of quality assurance and quality control apply but there are particular challenges associated with allergen mass spectrometry. The experimental design aimed at developing a new LC-MS/MS method is outlined showing the selection of protein and peptide targets, sample preparation, preliminary MS work-up, follow-up work preferably using an incurred material to assess peptide recovery before moving on to ‘real’ foods and validation.
In more detail, the protein molecular weight should be known, along with the full amino acid sequence and the protein should be unique to the foodstuff to be detected. At a minimum, the sequence should not be present in commonly analysed matrixes and other food ingredients. The protein expression should be characterised and preferably expression variability should be minimal. In other words, not altered to an appreciable extent by the tissue, species/cultivar, year/season, response to environmental/disease/stress factors, or post-harvest treatment. The protein should ideally not be subject to modification (either post-translational or processing-induced). If present, modifications must be considered when developing peptide targets and MS methods. The protein should not be subject to hydrolysis during storage or food processing and should be released from the matrix into the solution for detection. The protein should be reproducibly digested by the endoprotease enzyme used in the chosen sample preparation technique.
Data analysis is crucial in all analytical situations and although this aspect has similarities between ELISA and LC-MS/MS, in the latter, as a newer technique applied to allergen analysis, it is particularly challenging. The three essential components are: assurance around the detection of positive signals, calibration, and signal integration. Appropriate best practice is available (Johnson and Downs, 2019), along with transparent reporting criteria. These include full descriptions of:
- calibration material
- mathematical operations for calibration
- strategy used to select signals for use in quantification and those to be used qualitatively.
Adoption of these criteria will improve the evaluation, comparability, and transferability of LC-MS/MS methods.
An excellent example is the reference method for the determination of total allergenic protein content in processed food (milk in cookies) at clinically relevant concentrations, traceable to the SI and with well characterised uncertainty.
Food intolerance describes adverse reactions to a wide spectrum of non-protein compounds. Some typical examples are:
- histamine, annatto, tartrazine, sulphites, benzoic acid
- enzyme defects such as lactose intolerance
- short chain fermentable carbohydrates, FODMAPs, including fructo-oligosaccharides (fructans), galacto-oligosaccharides, fructose, sorbitol and mannitol.
Apart from FODMAPs, the compounds that give rise to food intolerance are mainly small molecules for which the difficulties of analysis exhibited by proteins are not encountered. Thus, HPLC and Ultra-Performance Liquid Chromatography (UPLC) methods, with suitable standards are readily available. For FODMAPs, relatively larger carbohydrate molecules High-Performance Anion Exchange chromatography with Pulsed Amperometric Detection is the most suitable approach. For sulphites a variety of methods are available including enzymatic analysis, ion-exclusion chromatography with amperometric detection, and variations on the Monier-Williams wet chemical reference method. It should be borne in mind that the Monier-Williams method has long been known to be interfered with positively by foods such as dried garlic and soya proteins. These foods typically assay around 200 mg per kg and 30 mg per kg as sulphur dioxide (SO2) respectively in the Monier-Williams method. Other foods such as onions and cabbage also typically give responses in the range of 3 – 20 mg per kg as SO2. These interferences are caused by the presence of volatile sulphur-containing compounds. In case of doubt, an LC-MS/MS should be applied.
To assist customers and especially risk assessors, laboratories must make analytical reports as informative as possible. Several elements must be included:
- method of analysis – stating whether it was ELISA, qPCR, or LC-MS/MS (or a combination), and additional information on the ELISA platform and nature of the calibrator should be encouraged
- it is vital that the unit of measurement is clearly reported, preferably as protein although some ELISAs report as the food commodity itself
- if the food commodity is the basis of the reported unit, the conversion factor for whole food to allergen protein must be given
- the unit should be specified every time the datum is cited in the report
- any suspected method cross reactivity if applicable
- recovery (if RM available, or by spiking) and whether or not the result is corrected for recovery
- measurement uncertainty.
The reporting laboratory should also be aware of and able to advise on the context, for example legislative requirements and thresholds.
These expectations do not come as cheaply as simply reporting a number and unit, owing to the added contextual awareness of food allergens that must be acquired and kept up to date.
- Abbott, M., Hayward, S., Ross, W., Godefroy, S.B., Ulberth, F., Van Hengel, A.J., Roberts, J., Akiyama, H., Popping, B., Yeung, J.M. and Wehling, P. (2010). Validation procedures for quantitative food allergen ELISA methods: community guidance and best practices. Journal of AOAC International, 93(2), pp.442-450. https://doi.org/10.1093/jaoac/93.2.442
- Allen, J. (1986) Immunoassays in food analysis. Nutrition Bulletin, 11(1), pp.46-54. https://onlinelibrary.wiley.com/doi/10.1111/j.1467-3010.1986.tb01224.x
- Allergen Bureau, Food Allergen Analysis. https://allergenbureau.net/food-allergens/food-allergen-analysis/, Accessed on 27 July 2022.
- AMC (2013) Technical Brief on PCR. https://www.rsc.org/images/TB%2059_tcm18-241515.pdf
- Breidbach A., Nørgaard J. V., Cubero-Leon, E. and Martinez Esteso, M. J. (2022) Assignment of a Reference Value of Total Cow’s Milk Protein Content in Baked Cookies Used in an Interlaboratory Comparison. Foods, 11(6), pp. 869. https://doi.org/10.3390/foods11060869
- Burns, M. (2019) A Perspective on Quantitative DNA Approaches. In: M. Burns, L. Foster and M. Walker, eds. DNA Techniques to Verify Food Authenticity: Applications in Food Fraud. pp 107-119. https://pubs.rsc.org/en/content/chapter/bk9781788011785-00107/978-1-7880...
- Food Allergy Research and Resource Program (FARRP) (2022). Food Allergens - International Regulatory Chart. University of Nebraska. https://farrp.unl.edu/IRChart
- Food Information to Consumers, Retained EU law 1169/2011. https://www.legislation.gov.uk/eur/2011/1169/introduction
- IFST (2021) Food Allergens Knowledge Hub. London: The Institute of Food Science and Technology. https://www.ifst.org/knowledge-hubs/food-allergens-knowledge-hub/guidance-consumers-food-allergens
- IFST (2021) Information statement on Food Allergy, London: The Institute of Food Science and Technology. https://www.ifst.org/resources/information-statements/food-allergy-0
- IFST (2021) Information statement on How to Choose and Instruct a Laboratory for Chemical Food Analysis. London: The Institute of Food Science and Technology. https://www.ifst.org/resources/information-statements/how-choose-and-instruct-laboratory-chemical-food-analysis
- IFST (2021) Information statement on Sampling for Food Analysis, London: The Institute of Food Science and Technology. https://www.ifst.org/resources/information-statements/sampling-food-analysis-key-considerations.
- Johnson, P. and Downs, M., 2019. From Signal to Analytical Reporting for Allergen Detection by Mass Spectrometry. Journal of AOAC INTERNATIONAL, 102(5), pp.1255-1262. https://doi.org/10.1093/jaoac/102.5.1255
- Lacorn, M., Lindeke, S., Siebeneicher, S. and Weiss, T. (2018) Commercial ELISA measurement of allergens and gluten: what we can learn from case studies. Journal of AOAC International, 101(1), pp.102-107. https://doi.org/10.5740/jaoacint.17-0399
- Remington, B. C.; Baumert, J.; Blom, M. W.; Bucchini, L.; Buck, N.; Crevel, R.; De Mooij, F.; Flanagan, S.; Stavropoulou, D.A.; van den Dungen, M. W.; van Ravenhorst, M.; Wang, S.; Walker, M. (2022) Practical Guidance on the Application of Food Allergen Quantitative Risk Assessment (QRA), Europe: Institute of Life Sciences- Europe. https://ilsi.eu/publication/practical-guidance-on-the-application-of-food-allergen-quantitative-risk-assessment-qra/].
- Rzychon, M., Brohée, M., Cordeiro, F., Haraszi, R., Ulberth, F. and O'Connor, G. (2017) The feasibility of harmonizing gluten ELISA measurements. Food Chemistry, 234, pp.144-154. https://dx.doi.org/10.1016%2Fj.foodchem.2017.04.092
- Walker, M., Burns, M., Quaglia, M., Nixon, G., Hopley, C., Gray, K., Moore, V., Singh, M. and Cowen, S., 2018. Almond or Mahaleb? Orthogonal Allergen Analysis During a Live Incident Investigation by ELISA, Molecular Biology, and Protein Mass Spectrometry. Journal of AOAC INTERNATIONAL, 101(1), pp.162-169. https://doi.org/10.5740/jaoacint.17-0405
- Walker, M.J. (2018). Food Allergens: An Update on Analytical Methods. In: Melton, L., Shahidi F., Varelis, P. (eds.). Encyclopaedia of Food Chemistry, 1st ed, pp. 622-639. Elsevier. https://www.elsevier.com/books/encyclopedia-of-food-chemistry/varelis/978-0-12-814026-0
Institute of Food Science & Technology has authorised the publication of the following Information Statement on Allergen Analysis – key considerations (including gluten & food intolerance compounds).
This Information Statement has been prepared by Dr Michael Walker FIFST, peer-reviewed, and approved by the IFST Scientific Committee.
This information statement is dated August 2022.
Michael Walker acknowledges the reuse of certain content that was compiled for an eSeminar (link to follow when available) on this subject for the Joint Knowledge Transfer Framework for Foods Standards and Food Safety Analysis, funded by the Department for Environmental and Rural Affairs (Defra), the Food Standards Agency (FSA), Food Standards Scotland (FSS), and the Government Chemist (funded by BEIS, the Department for Business, Energy and Industrial Strategy).
Michael Walker acknowledges benefitting from funding via LGC for the preparation of the peanut, milk, egg white, almond, hazelnut, and walnut reference materials, preparation of the FSA-funded e-seminar on allergen analysis, and payment from LGG Standards for an e-seminar on allergen analysis.
Professor Katrina Campbell, Queen's University Belfast, is thanked for providing content on the topic of biosensors.
The Institute takes every possible care in compiling, preparing, and issuing the information contained in IFST Information Statements, but can accept no liability whatsoever in connection with them. Nothing in them should be construed as absolving anyone from complying with legal requirements. They are provided for general information and guidance and to express expert professional interpretation and opinion, on important food-related issues.